Hearing aids are categorized as Class 1 or Class 2 medical devices by the Food and Drug Administration. The large majority of hearing aids are in the Class 1 category, which means they are exempted from the premarket notification and FDA clearance requirement. Hearing aid manufacturers are required to register their establishments and bring their products into compliance with a number of “recognized consensus standards” developed and published by the American National Standards Institute (ANSI).[1]
The state of affairs described above has not always been the case in the history of hearing aids. Hearing aids were not considered medical devices until 1977 and recent changes in FDA thinking suggest they will not be considered medical devices in the near future.[2] This is the first of several posts reviewing ways in which technological advances, interest groups, economics, and politics have shaped the definition and regulatory treatment of hearing aids at different times in their history.
Early History of Hearing Aids
Ear Resonators [3]
The idea of boosting sound intensity to the ear is as old as cupping a hand behind the ear to improve hearing in noisy backgrounds or for sounds at a distance. Ear cupping provides about 10 dB of passive amplification in the mid-frequency range, and thus qualifies hand cupped qualifies as the first hearing aid. This “handy” hearing aid is used by everyone, although those with hearing loss resort to it more regularly, so it is functions as a hearing enhancer rather than a treatment for a medical condition.
Speaking tubes (Fig. 1) were another early means of channeling and conveying sound to the ear. These devices, often hand-made on the spot, offered the additional feature of enabling private communication in and across public places, which makes them somewhat analogous to modern-day telephones. Indeed, the first speaking tubes were created in Puritan times to allow courtship communication in public.[4] Thus, speaking tubes were an early form of hearing aid, used as hearing enhancers by people with normal and impaired hearing.

Figures 2. Ear Trumpet
Ear trumpets (Fig. 2) were a technological improvement that combined ear cupping with extended tubes to passively amplify sound by capitalizing on resonant frequencies determined by the dimensions of the trumpet. Trumpets could boost mid-frequencies in a narrow range by 10-20 dB. They had the added feature of directionality, since the “wearer” could direct the trumpet toward the sound source. But, they were cumbersome and unsightly to the point that only those with hearing loss chose to use them. As a result, ear trumpets were the first hearing aid devices designed to treat a disability problem rather than enhance (normal) personal hearing.
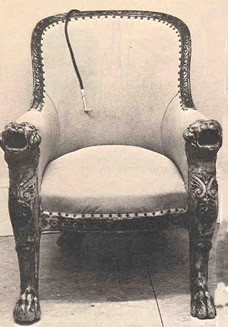
Figure 3. Acoustic throne.
Ear trumpets were in vogue throughout the 18th and 19th centuries, and assumed a variety of designs that aimed to conceal the user’s disability. The most famous device was the acoustic throne of King Goa of Portugal, shown in Fig. 3. It was not wearable and it required speakers to kneel and speak into the chair’s arms, which concealed resonator cavities and a tube in back. Those features were not a problem for a king, but posed problems for others with hearing difficulty who could not control their communication situations as fully. They needed a hearing aid they could wear that was portable.

Figure 4. Binaural horn device.
Wearability, plus the considerable technological advance of binaural input, was captured by the beard receptacle design shown in Fig. 4, designed to be worn under a man’s beard or a woman’s scarf.
Tune in for part 2 of this series, where we’ll see how electronic advances introduced not only the modern hearing aid, but manufacturing, business, and government regulation.
Editor’s Note: This is the first post in a 7-part series. Click link for Part 2.







hi, superb blog page, and an excellent understand! at least one for my book marks.
Wow — thanks for the kind words. We’ll try to keep up the good work!