In 1993 the American Academy of Audiology (AAA) held their 5th annual convention in the Arizona desert. The official attendance for the meeting was reported to be 2,300. Move the clock 23 years and a new generation of audiologists joined a stalwart group of AAA founders, exhibitors, clinicians and countless others – some 6,000 attendees in total – to celebrate audiology.
 Given the hundreds of learning labs, exhibitor courses, featured sessions and scientific podium presentations, there is much to celebrate – audiology has never had a more dynamic & diverse presence in academia, industry, healthcare and commercial business.
Given the hundreds of learning labs, exhibitor courses, featured sessions and scientific podium presentations, there is much to celebrate – audiology has never had a more dynamic & diverse presence in academia, industry, healthcare and commercial business.
Yet, in conversation with some conference-goers, a pall may have been cast over this meeting.
Domino Effect
Although more than 170 exhibitors showcased the latest technology in the profession, several industry behemoths were conspicuously absence from the expo floor. This list includes Starkey and Amplifon, who have not exhibited in a few years. In 2016, they were joined by GN Resound and Unitron.
According to sources who wish to remain anonymous, other key industry players are contemplating a similar strategic move and leaving their exhibit space in 2017.
Even though Oticon, Signia and Widex had busy booths this year, the withdrawal of major industry players may signal a domino effect in how industry incumbents market their new products, perhaps now preferring a more direct, local approach with their customers, rather than the large splash coinciding with the annual AudiologyNow event.
Looming Changes Ahead?
In addition to the absence of key players on the show floor, several sessions alluded to the impact of the PCAST’s recommendations to create a new, Basic category of direct-to-consumer hearing aids (along with three other recommendations that could essentially deregulate the industry) might have on the profession of audiology.
In what is sure to be an arduous process, the FDA is scheduled to hold on April 21st, a full day workshop: Streamlining Good Manufacturing Practices (GMPs) for Hearing Aids.
Thursday’s conference will be simulcast at https://collaboration.fda.gov/gmp042116/. Readers are encouraged to view the session in real-time.
Déjà vu All Over Again

David Kessler
Veteran audiologists may recall that the 5th Annual AAA convention in Phoenix occurred a few months before another pivotal US Government meeting concerning the regulation of hearing aids.
It was in 1993 that then FDA Commissioner David Kessler spearheaded a congressional inquiry that lead to significant changes in hearing aid product claims. A transcript of the September, 1993 congressional meeting involving Dr. Kessler, along with several witnesses from ASHA, IHS and AAO (apparently AAA was too young of a group to be invited) participated in the proceedings.
Reading the transcripts from the 1993 proceedings is a reminder that many of the key issues audiologists and other stakeholders currently face have remained largely unchanged.
Below is the agenda for the April 21st meeting.
.
GMPs for Hearing Aids Agenda

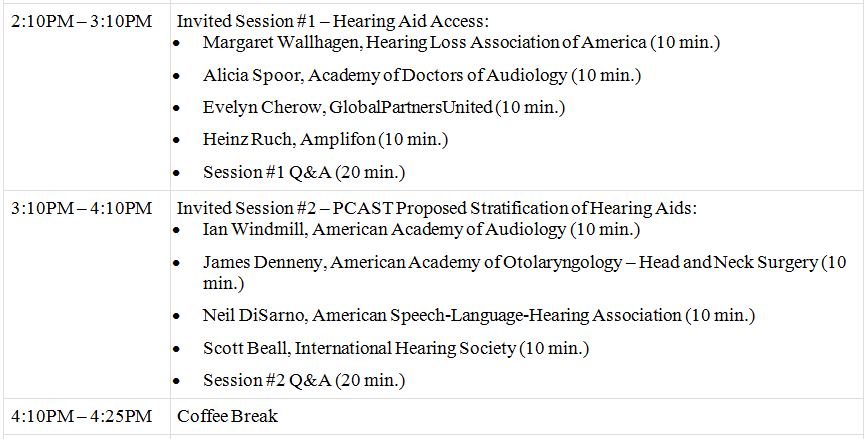
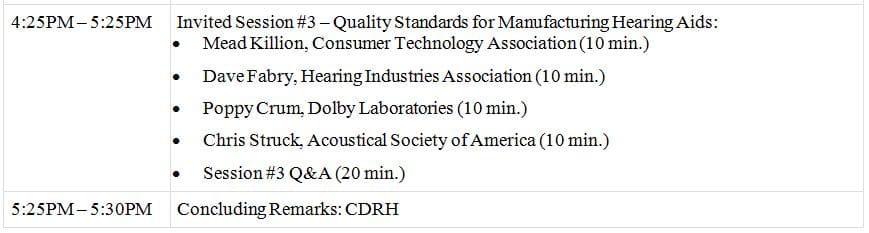
*title image courtesy consumerproductmatters








Curious as to whether cost of the show ( and the pressure to have a fancy, multi level display, )a costly event coinciding with the show is the reason for so many companies pulling out, or if it is truly a desire on these companies parts to do more local
Marketing .
Actually, Dr. Taylor….although you were likely far too young to know, during that 1993 FDA update of the 1977 Hearing Aid Regulations, AAA was indeed present and involved (see AT, 5:6, pg 22). It was a preliminary meeting of the US Senate Committee on Aging, but it was during Dr. Lucille Beck’s presidency and several of the AAA Board members collectively drew up testimony to put into record the crucial role audiologists play in the hearing aid process — and I was selected to deliver the presentation. As an aside to that story, later that week while in Wash. DC, Lu and I were getting a breakfast bagel and ran into Commissioner David Kessler standing in line with us! We managed to get in a few more words about audiology directly to him while he was putting cream cheese on his bagel!
Thanks for the clarification, Dr. Northern. I thought that even at an early age AAA was involved in such matters, I just couldn’t find any documentation with respect to the meeting I cited. Dr. Kessler, by the way, has a great new book, called Capture: Unraveling the Mystery of Mental Suffering
As a consumer with hearing loss, I would like to see some of this hearing aid panel and ask that captions be included on the simulcast.
Thank you.
Amen sister! We “defies” are tired of all those in the hearing healthcare sector continuing to post videos without captioning.
Are Starkey, GN Resound and Unitron present at other shows in North America? Maybe it’s just THIS show that is shrinking?
The number of participants from 1993, I assume George Frye was there, and 2016 is significant. Another number to look at is the international participation. Making the event accessible to the Audiology Industry professionals from around the Globe is what will continue to grow the show. Audiology NOW needs to compete with EUHA for Exhibitors and Participants.
I attended the Phoenix exhibition. This is really a story of the Rise and Fall of AAA. The last show I attended was several years ago. The exhibit hall then was amazing. Not so this year. What a change. Attendance is way down compared to years ago. The focus of the last show I attended was Ethics. AAA was preaching how awful it is that audiologist take bags from vendors. This was the beginning of the downfall. There are so many important issues facing our profession, yet AAA chose to focus on bags and pens. The vendors are the backbone of our industry, and the main financial support for AAA. AAA needs to really examine what they are doing to promote audiology as a profession. What is AAA doing to fight PSAPs. What is AAA doing to promote direct access. What is AAA doing to stop hearing aids sold online by United HC? Etc etc. Time for AAA to do some serious soul searching.