The zinc-air hearing aid battery (cell) of today has small holes in it. Why is this?
Background
Hearing aid batteries currently are identified as zinc-air batteries (in hearing aids, actually a cell rather than a series of cells comprising a battery). The use of zinc-air cells for hearing aids was mandated in the U.S. by Big Brother in Washington, D.C. in 1996.
Prior to zinc-air cells, mercury was used in hearing aid cells as one of the most common active ingredients, although a few of the larger sized cells used silver as the active ingredient.
However, in 1996 the Mercury-Containing and Rechargeable Battery Management Act was enacted to facilitate the recycling of nickel-cadmium (Ni-Cd) and certain small sealed lead-acid (SSLA) rechargeable batteries and to phase out the use of mercury in batteries. The reason for a phase out program existed because a problem arose in that the EPA (Environmental Protection Agency) action failed to take into account that hearing aid cells did not have available an alternate mercury-free cell design. Having this brought to their attention, they provided an exemption from this ban for button cells used in watches and hearing aids until such time that a robust mercury free design was available.
Figure 1. Packages of sample 675 size zinc-air cells provided to hearing aid manufacturers to promote these new products. Manufacturers included such samples in the hearing aid package to the client. Note: “Not for resale.”
Fortunately, battery companies had already been experimenting with and providing zinc-air cells since about 1977. Zinc-air cells are metal-air cells powered by oxidizing zinc with oxygen from the air. The development goal was to provide a cell that had a longer and constant performance and was not initially an attempt to eliminate mercury to meet some future requirement. It was the cell life performance and not a mercury issue that battery companies were excited about – excited to the point of providing samples to hearing aid manufactures to include in the hearing aid package as part of their promotional activities (Figure 1). Not only did zinc-air cells provide a longer and more stable battery life (Figure 2), they offered other advantages, and a few disadvantages as shown in Figure 3. At the time, most hearing aids operated using size 675 cells.
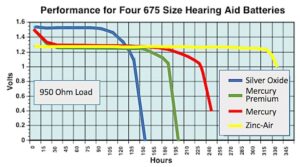
Figure 2. Hearing aid battery discharge rate in hours showing the comparison of the impact on battery life among the batteries at the time when zinc-air cells were introduced. Except for hours of life, other size cells perform similarly.
In 2006, the National Electrical Manufacturer’s Association (NEMA) asked all battery companies to remove mercury from hearing aid cells by June 30, 2011. Leading battery companies had already been working toward this and voluntarily agreed to the goal, and currently, zinc-air cells used in hearing aids are mercury free. Still, it is noteworthy that zinc-air cells all included mercury as an active ingredient. This practice of zinc-air cells containing some mercury continued at least ten years following the EPA Act and up until the NEMA request, or for a total of almost thirty-five years following the introduction of the first zinc-air cells! Progress does not always occur rapidly.
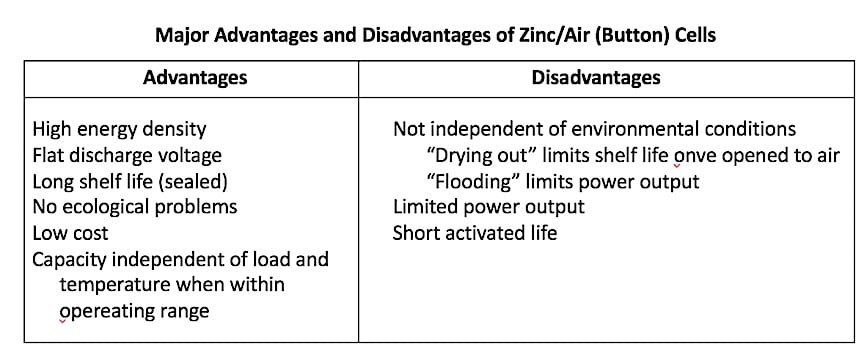
Figure 3. Major advantages and disadvantages of zinc-air button hearing aid cells.
An advantage of mercury-free hearing aid cells is that they require no special handling when being discarded or recycled.
But, What Does This All Have to Do with Those Little Holes in the Batteries?
For increased battery life, the energy density within the cell had to be increased. This was done by replacing most of the mercury with zinc, and when combined with air, produces the greatest energy density. If the air is not stored in the cell, even greater space can be used for the zinc and other chemicals to ensure the stability of the cell.
Any attempt to store air in such a small sized cell would neither be economical nor practical. As a result, the air to create the chemical reaction would come from outside the cell, and this is the role of the holes – to allow air to enter. But, it had to be kept from the cell until the time of need. Hence, the adhesive tab seal that is removed to activate the cell. Essentially nothing happens until the tab is removed, but once removed, the process cannot be reversed. A typical sealant is polyester tape.

Figure 4. Five air ports are shown in this 675 size hearing aid cell.
In reality, the role of the air holes is important because the instantaneous current is limited by the number and size of the air holes (Figure 4). High pulsed inputs (which hearing aids are exposed to) cause the current to rise and fall (incoming sound is not steady).
If the cell cannot supply enough current to the amplifier during the peaks, distortion results. My best explanation has been to describe this like a pump sucking air. In fact, in the earlier days of zinc-air cells, a consistent problem for returned hearing aids for repair/credit was that they sounded distorted or cut in and out at times. This was more than likely the result of the zinc-air cell not performing properly rather than a problem with the hearing aid circuitry. It was known in the industry that about the only way to check this out was to insert a mercury cell and see if the same thing happened. This usually solved the problem and any number of individuals made the trip back to mercury cells during that transition period.
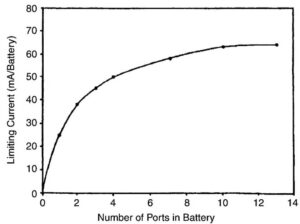
Figure 5. This graph shows the relationship between the number of ports (air holes in a 675 size cell) and the limiting current in mA. The limiting current increases with increased air access. This is a reason why larger-sized cells have a greater number of holes.
Depending on the brand and size hearing aid cell used, the number of holes is likely to vary, even among manufacturers. Figure 5 is a graph showing the relationship between the number of ports (air holes in a 675 size cell) and the limiting current in mA. The limiting current increases with increased air access. As a result, larger sized zinc-air cells will have the most holes because the larger size cell suggests a stronger hearing aid, which means that a higher storage capacity is needed, and greater air activation will be required to allow the aid to manage the higher current drain required. The number of air holes runs from about one to five, depending on the cell size and manufacturer.
It is important to note that the number and size of holes has to be controlled in order to maximize the chemical reaction of the cell ingredients. Too much, or not enough oxygen results in less than satisfactory results.
Zinc Air Batteries and the Future of Hearing Aid Battery Technology
The demand to design zinc-air cells to meet greater power and energy needs of increasingly more complex and demanding hearing aids is something that hearing aid battery manufacturers are currently facing. Will future power suppliers continue to offer zinc-air or some alternative?
Some changes can be expected to happen, but we already have been seeing in recent years the shift to rechargeability, with silver-zinc and, now, many manufacturers moving towards Lithium Ion as the most practical alternative to meet greater demands of today’s hearing aids. Companies like Widex, for example, have also introduced fuel cell technology into hearing aids, as an alternative to zinc-air cells, which will be another interesting development to watch.







