Part II – XP Peritympanic Impact Leading to the CIC Hearing Aid
This post is a continuation of a blog from a couple of weeks ago describing the road to the development of the CIC (completely-in-the-canal) hearing aid. What started as an effort to reduce the occlusion effect for ITC (in-the-canal) style hearing aids, led to the development of a dramatic application of custom-molded hearing aids – the peritympanic hearing aid. That development, in turn, became the primary driver to the CIC hearing aid.
True Deep Canal Hearing Aid Fittings Identified as Part of a Manufacturer’s Product Offering
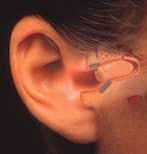
Figure 1. The Philips XP Peritympanic hearing instrument. Its positioning deep within the external auditory canal and in close proximity to the tympanic membrane made it essentially invisible, but even more, offered significant acoustic benefits.
In mid 1991, this author was contacted by Philips Hearing Instruments of the Netherlands and was hired as a consultant to a new product they intended to introduce, the XP Peritympanic (Figure 1), a hearing instrument placed totally within the ear canal and in close proximity to the tympanic membrane. Philips’ interest in the author was based on his experience with the deep canal ITC instruments reported early in 1991 by Staab and Finlay{{1}}[[1]]Staab, W.J., and Finlay, B. A fitting rationale for deep fitting canal hearing instruments, Hearing Instruments, Vol. 42, No. 1, pp 6, 8, 10, 48, 1991[[1]].
The interest by Philips to place a product totally within the ear canal and in close proximity to the tympanic membrane (eardrum) was based on expected acoustical benefits, and also driven by two medical events at the time, both of which aimed to improve hearing quality and device invisibility, with both focused on invasive surgical techniques.
In Japan, interest was focused on totally and impartially implantable devices. These used a piezoelectric crystal fixed to the stapes in the middle ear and was powered by an induction coil fitted subcutaneously. Depending on whether the instrument was totally or impartially implanted, other components, such as secondary induction coil, microphone amplifier, and batteries were either also implanted subcutaneously or located in a contour of the ear. When the batteries of this type instrument would run flat, the user would connect via an induction coil to a battery charger for re-charging.
In the United States, another group used electromagnetic induction by ossicular transduction. This used a magnet in the ossicular structure placed in such a way that it faced as closely as possible, and aligned as perfectly as possible, with an induction coil. When transversed by an electrical current, the coil vibrated the magnet and transmitted a signal to the oval window via the ossicular structure.
Philips recognized that while both these approaches provided significant benefits, they required surgery that could cause inconvenience, scar tissue, and psychological discomfort. And, in the case of the electro-magnetic system, the scar tissue after surgical intervention could cause the auditory canal to become narrower than the original auditory canal.
Instead, Philips decided to develop a hearing aid that fit very deeply in the ear canal to take the best advantages of a small residual ear canal volume. It was called the Philips XP Peritympanic hearing aid. In developing the first commercial deep canal hearing aid, Philips set a mission to design it by satisfying the needs for exceptional acoustic performance and invisibility, but without invasive surgical methods. Additional design features involved: 1) using only the natural space within the ear canal, and 2) positioning the instrument deeply within the bony portion of the ear canal, and 3) reducing the residual volume between the end of the instrument and the tympanic membrane to a very small minimum.
XP Peritympanic
The Philips XP Peritympanic hearing aid was intended specifically for deep canal fittings and engaged dispenser involvement in patient preparation methods never before practiced when fitting hearing aids. This was based around taking an ear impression of the entire ear canal, including an imprint of the tympanic membrane (Figure 2). Ear impressions had never been made this deeply previously, at least not intentionally. Special training was required for the taking of ear impressions, along with proper tools and materials for this to occur. The final fitting rationale and test results were published in 1992{{2}}[[2]]Staab, W.J. The XP Peritympanic instrument: fitting rationale and test results, The Hearing Journal, Vol. 45, No. 10, pp 21-26, 1992[[2]].
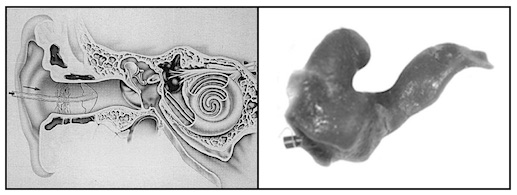
Figure 2. XP ear impression procedure. A specially designed silicone rubber aerating tube is inserted into the ear canal until it comes into contact with, and follows the form of the tympanic membrane (left). A specially-formulated ear impression material is then injected into the ear canal to obtain an impression of the entire ear canal, including an imprint of the tympanic membrane (right).
The Philips XP Peritympanic hearing instrument introduced a dramatic approach to fitting a product into the ear canal. Initial comments by “experienced” audiologists (often by those who were not fitting hearing aids) was that this was dangerous and would never be acceptable – the ear canal was too “delicate” to be fooled around with and should not have an ear impression taken, or a product placed as proposed. (Forward to 2015 in which similar products are being fitted, which emphasizes that initial reactions are not always made based on knowledge or experience).
In spite of these dire warnings from some in the dispensing communities, consumer interest was very high. Because of this interest, many dispensers (audiologists and traditional) whose livelihood depended on fitting hearing aids, requested for and engaged in programs which were conducted world-wide in order to provide the necessary training to take deep impressions and learn how to fit such products.
Interestingly, major problems encountered ended up not related primarily to the “dire warnings” of ear canal damage, but to the size of components (microphones, speakers, cell, amplifier) that often could not be fitted into small ear canal volumes to the depth required, and therefore fulfill the requirement for concealment within the ear canal.
Still, many dispensers were uncomfortable with taking ear impressions to the tympanic membrane, and competitor manufacturers sought ways to capitalize on the heightened interest created by this new hearing aid style by “modifying” the impression-making process, the hearing aid design, and location within the ear canal.
This paper trail discussion leading to the origin of the CIC hearing aid will continue in a following post.