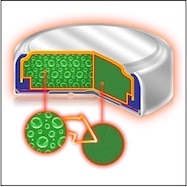
Hearing aids are electronic devices that apply amplification provided by a power supply, most usually called the “battery.” The power supply provides the energy to drive the hearing aid (an electrical load). What the power supply (hearing aid battery) does is to essentially convert one form of electrical energy to another.
Battery Basics for Hearing Aids
Although historically, “battery” refers to a power supply composed of multiple cells, the usage has evolved to include power supplies composed of a single cell, as used in most hearing aids.
Primary (Disposable) Battery
These are single-use, or disposable batteries, used once, and then discarded. They are most often used in portable devices that have low current drain, such as hearing aids or in other situations where alternate electrical power is intermittently available. The electrode materials are essentially irreversibly changed during discharge, meaning that they cannot be readily recharged, or returned to their original forms.
Prior to current zinc-air cells for hearing aids, older mercury disposable cells could typically lose 8 to 20% of their original charge when stored at room temperature (20-300 C) for the first month, and then 2% for subsequent months1. It was because of this that some companies recommended storage in a cooler environment (refrigerator, not freezer). However, with zinc-air cells used in hearing aids this is no longer an issue and storage in a refrigerator is no longer recommended.
Secondary (Rechargeable) Battery
A secondary battery can be discharged and recharged multiple times, with recharging provided by an alternating current (AC) electrical power supply (wall socket). The electrode’s original composition can be restored by reverse current. Some hearing aids provide for the interchange of use by secondary and/or primary batteries. However, secondary batteries cannot be recharged indefinitely. This is because they lose electrolyte, dissipate the active materials, and show sign of internal corrosion. Secondary batteries have a more rapid self-discharge rate (loss of original charge) than do primary batteries.

Figure 1. Miracle Ear Charger, Dahlberg Electronics, Inc., Hearing Systems Division. The hearing aid(s) turned off, but are left in the hearing aid with both placed in the round cavity. Batteries were to be charged daily for 8 hours minimum to ensure adequate charge. (Staab collection, product of the late 1960s or early 1970s).
The hearing aid industry has made available rechargeable batteries for many years. In the 1960s and 1970s, especially internationally, rechargeable cells were used commonly. These were recharged by taking energy from a larger milliamp capacity and transferring it to the hearing aid button power supply via direct usurpation (DK10 and DK20 cells). Others have used solar power to restore battery function. However, the life cycle performance and recharging time of earlier products were not good. The past few years have fostered a renewed interest in rechargeable batteries, partially as a result of the use of wireless hearing aid products/features that require higher current drain2. These have contributed to shorter battery life. This author has written previously about the potential future use of body energy to power hearing aids.
Currently, a number of rechargeable hearing aids are available. These consist mostly of hearing aids that are placed in a charger, with the cell charging while remaining in the hearing aid. These are a takeoff on rechargeable hearing aids sold by Miracle Ear (Figure 1) and a few others, mostly in the early 1970’s, but with improved cells and ability to hold the milliamp capacity.
Additionally, a survey conducted by Kochkin in 2008 showed that 45% of consumers rated battery life of their hearing aids to be less than satisfactory. They believed that rechargeable hearing aids (actually rechargeable batteries) would increase the likelihood of new purchases3. This request was expressed again in 20144.
Battery Capacity
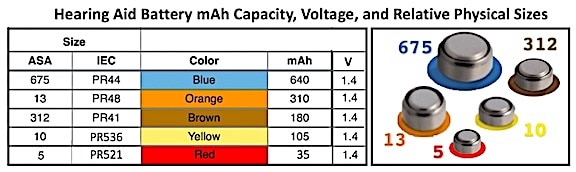
This refers to the amount of electric charge the battery can deliver at the rated voltage. In hearing aids, the size of the battery generally determines what the battery capacity is. The more electrode material contained in the cell, the greater its capacity. Consider an analogy of the battery as a water storage tank. The larger the tank, the more water it can store. In a hearing aid battery, the larger the battery, the greater the mAh (milli Amp. hour is the measurement unit) capacity (storage) of cells used in hearing aids, even though large and small hearing aid cells develop the same open-circuit voltage. In the case of hearing aids, the open-circuit voltage (V) is 1.4 Volts (Table 1). It is important to understand that the mAh capacity varies among manufacturers. The values shown in Table I are an approximate estimate from among the suppliers and shows the common hearing aid battery sizes. The size identifications are those designated by the ASA (American Standards Institute) and IEC (International Electrotechnical Commission). Some companies often use a different letter(s) in front of the numbers, but all use the color combination for proper identification.
Table 1
Battery Life
Generally, battery life (hours of continuous potential use) is calculated based on the current rating mAh. Ampere is an electrical unit used to measure the current flow towards the load – in this case, the hearing aid amplifier. The battery life can be calculated from the input rating of the battery (mAh) and the load current (current drain) of the circuit. Battery life will be high when the load current is less and vice versa. The calculation to determine the capacity of a battery life in hours can be mathematically derived from the formula shown here:
In making the calculation, the multiplier of 0.7 is not often used to calculate hearing aid battery life because the zinc-air cells used are believed to hold their storage capacity for a long period of time without significant deterioration. In that case, battery life calculations will be longer. However, it may apply to rechargeable hearing aid batteries. A more detailed discussion of issues that affect battery life is found in previous posts (February 9, 16, 23, and March 1, 2016).
The percentage or fraction of the stored charge a battery can deliver depends on many factors, among the most important are: battery chemistry, the current drain (rate at which the charge is delivered), the required voltage required of the hearing aid, how long it has been stored, and the ambient temperature. At low temperatures, a battery cannot deliver as much power because its capacity drops.
Electrode Materials
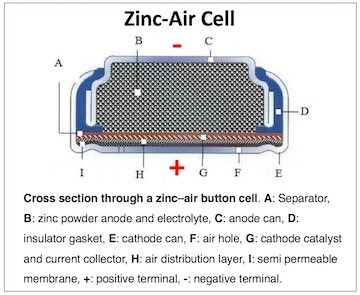
An electrolyte substance within the battery (B) allows chemical reactions (ion movement) to be completed between the anode (negative side – C ) and cathode (positive side – E), allowing current to flow from the battery and perform work (Figure 2). In the case of hearing aids, the work is amplification.
Brief Hearing Aid Battery History
The very first hearing aids were powered by Leclanche batteries. Originally a wet cell (cell having a liquid electrolyte), it was adapted to operate as a dry cell (cell using a past electrolyte, with only enough moisture to allow current flow). This allowed for operation in any orientation without spilling, and making it suitable for portable equipment, such as hearing aids.
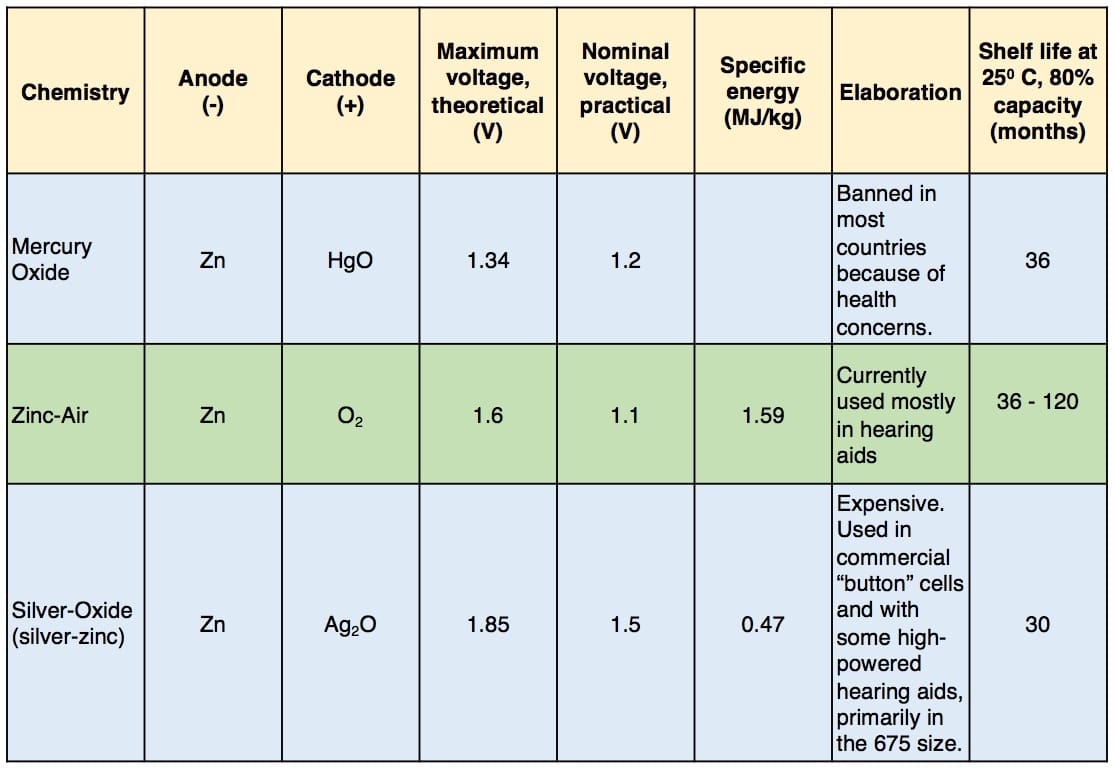
Many types of electrochemical cells have been produced using various chemical processes and designs. These include galvanic cells, electrolytic cells, fuel cells, and voltaic piles5. The most common for hearing aids have been electrolytic cells, but some hearing aid companies are preparing for fuel cells (more on this later). Table II shows the most common hearing aid cell type chemistries that have been used in the “modern” era, along with additional descriptive information.
Table II. Hearing Aid Battery Chemistry
 Key: Zn = Zinc; HgO = Mercury Oxide; O2 = Oxygen; Ag2O = Silver Oxide
Key: Zn = Zinc; HgO = Mercury Oxide; O2 = Oxygen; Ag2O = Silver Oxide
Current hearing aid use mostly the zinc-air cell. The primary reason for this relates to the 1996 Mercury-Containing and Rechargeable Battery Management Act that was enacted to facilitate the recycling of nickel-cadmium (Ni-Cd) and certain small sealed lead-acid (SSLA) rechargeable batteries, and to phase out the use of mercury in batteries. Unfortunately, the EPA (Environmental Protection Agency) action failed to take into account that hearing aid cells did not have available an alternate mercury-free cell design. Having this brought to their attention, the EPA provided an exemption from this ban for button cells used in watches and hearing aids until such time that a robust mercury free design was available. Progress did not occur rapidly in that it took a total of almost thirty-five years following the introduction of the first zinc-air cells to make this transition!
Next Post: Fuel Cells for Hearing Aids?
Featured image courtesy of Energizer.
References
- Self-discharge of batteries – Corrosion Doctors Retrieved 27 December 2016.
- Freeman, B. A new door to rechargeable hearing aid battery solutions. Hearing Review, July 29, 2015.
- Kochkin S. MarkeTrak VIII: The key influencing factors in hearing aid purchase intent. Hearing Review. 2012;19(03):12-25.
- Better Hearing Institute (BHI). MarkeTrak 9. Washington, DC: BHI;2015.
- Spotlight on Photovoltaics & Fuel Cells: A Web-based Study & Comparison (PDF). pp. 1–2. . Retrieved 24 December 2016.